The Buffalo Neuroimaging Analysis Center (BNAC) is a dedicated research center that has expertise in developing innovative MRI techniques and methods through a multi-disciplinary team of MR physicists, computer scientists, and clinical neuroimagers. By leveraging new advances in machine learning and artificial intelligence (AI), BNAC has been able to develop numerous new MRI acquisition and analysis methods that are capable of analyzing large-scale datasets with tens of thousands of scans across multiple timepoints in thousands of individuals.
Deep learning is a state-of-the-art technique for developing computer vision methods capable of rivaling or even exceeding human-level ability in specific areas. In addition to the development of new MRI techniques, BNAC has also been working to integrate deep learning systems into research workflows to improve overall study quality and precision. Unlike most research datasets, which are often curated by single investigators (or even trainees), BNAC uses a three-level quality control system where scans are reviewed by multiple evaluators and resulting gold-standard ROIs are painstakingly edited to ensure the highest possible accuracy. This in turn provides unparalleled training data for us to teach AI systems to identify and delineate brain regions and areas of pathology. By feeding this back into clinical trial activities, we can augment human operators to further improve precision of lesion delineation and accuracy of lesion detection.
Another highlight of BNAC’s AI activities includes the development of a translational tool to measure thalamic volume on real-world clinical images. The thalamus is involved in many neurological functions and is a critical structure for examining neurodegeneration in MS and other diseases. BNAC developed a deep-learning method called DeepGRAI (Deep Gray Rating via Artificial Intelligence) for measurement of this important biomarker on conventional T2-FLAIR MR images, while providing accuracy rivaling the best research-quality sequences.
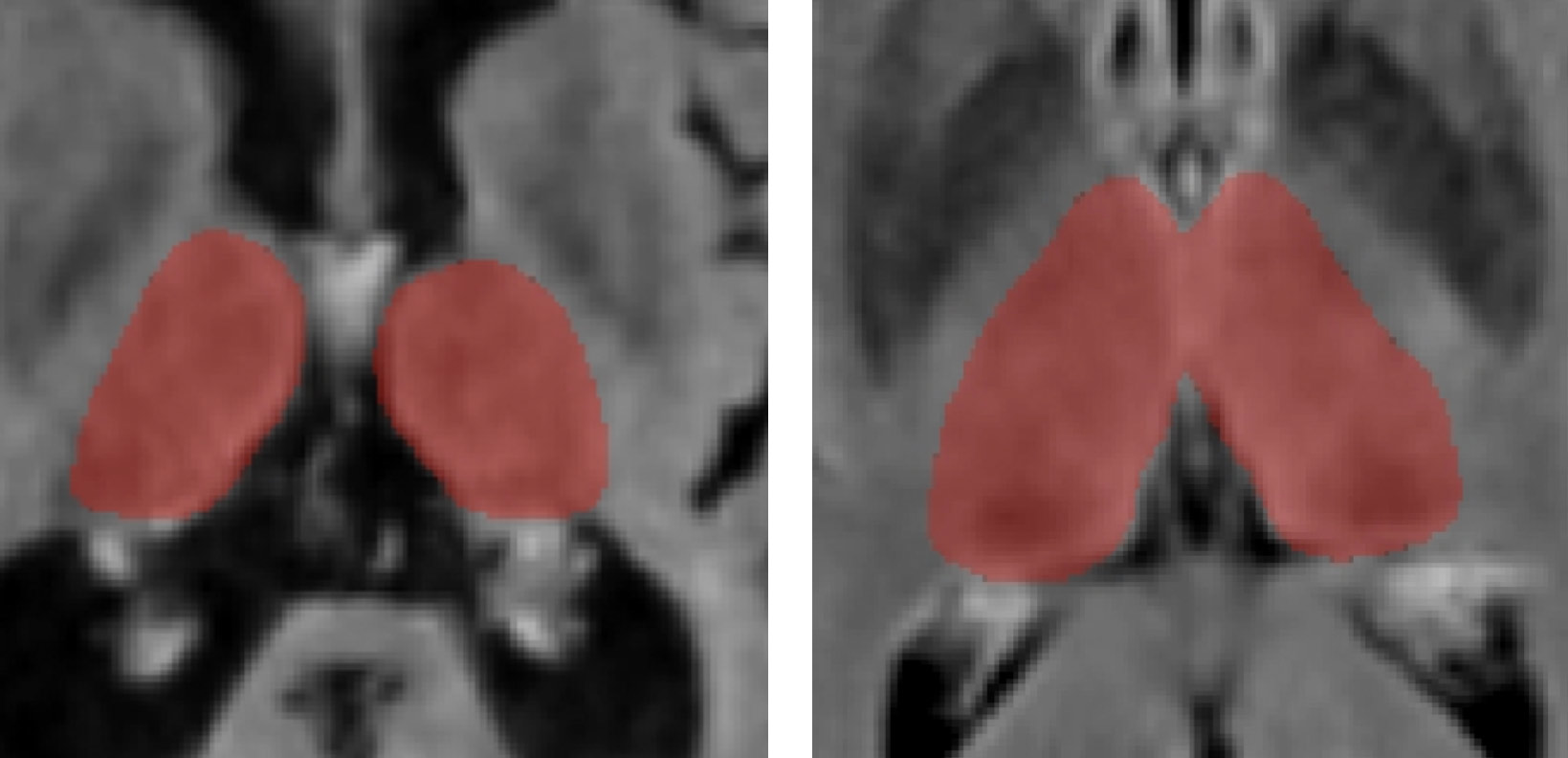
Results of DeepGRAI segmentation algorithm on cases with very high (left) and low (right) levels of atrophy. The classifier performs on clinical routine T2-FLAIR images well in both cases despite substantial differences in brain morphology and lesion volume.
For more information about clinical and preclinical imaging endpoints, click here.
FEATURED RECENT INNOVATIVE MRI TECHNIQUES/AI STUDIES
- Zivadinov R, Bergsland N, Jakimovski D, Bartnik A, Schweser F, Eckert S, Hojnacki D, Weinstock-Guttman B, Dwyer MG. Neuromaging phenotype characterization of aggressive multiple sclerosis: A case-control study. Brain Comm 2025;7(4):fcaf254.
- Dwyer MG, Bergsland N, Bartnik A, Jakimovski D, Noteboom S, Schoonheim M, Steenwijk M, Pei J, Clayton D, Zivadinov R. Quantifying cortical lesions in large legacy multiple sclerosis clinical trial MRI datasets using multi-contrast post-processing and deep learning. medRxiv 2025.04.08.25325461;doi: [Open Article]
- Dwyer MG, Pei J, Clayton D, Goldman D, Cabatingan M, Bartnik A, Jakimovski D, Bergsland N, Zivadinov R. The impact of ocrelizumab on cortical lesion accrual in primary progressive MS in the double-blind, phase 3 ORATORIO study.
- Zivadinov R, Bergsland N, Jakimovski D, Weinstock-Guttman B, Lorefice L, Schoonheim MM, Morrow SA, Picone MA, Pardo G, Zarif M, Gudesblatt M, Nicholas JA, Smith A, Hunter S, Newman S, AbdelRazek MA, Hoti, I, Riolo J, Silva D, Fuchs TA, Dwyer MG, Benedict RHB. Thalamic atrophy and dysconnectivity are associated with cognitive impairment in a multi-center, clinical routine, real-word study of people with multiple sclerosis. Neuroimage Clin 2024;42:103609 [Open Article]
Contact Our Group Today
Part of BNAC’s mission is to help share our tools and experience with our colleagues and other industry partners. If you need help with advanced MRI analysis techniques and AI in your research or clinical trial work, please reach out to discuss how we can assist. Our group brings decades of experience and expertise to every collaborative study and service partnership.

